Nano Engineering of Laser Induced Graphene for Next-generation Batteries
Highly Doped 3D Graphene Na-Ion Battery Anode by Laser Scribing Polyimide Films in Nitrogen AmbientConventional graphite anodes can hardly intercalate sodium (Na) ions, which poses a serious challenge for developing Na-ion batteries. This study details a novel method that involves single-step laser-based transformation of urea-containing polyimide into an expanded 3D graphene anode, with simultaneous doping of high concentrations of nitrogen (≈13 at%). The versatile nature of this laser-scribing approach enables direct bonding of the 3D graphene anode to the current collectors without the need for binders or conductive additives, which presents a clear advantage over chemical or hydrothermal methods. It is shown that these conductive and expanded 3D graphene structures perform exceptionally well as anodes for Na-ion batteries. Specifically, an initial coulombic efficiency (CE) up to 74% is achieved, which exceeds that of most reported carbonaceous anodes, such as hard carbon and soft carbon. In addition, Na-ion capacity up to 425 mAh g−1 at 0.1 A g−1 has been achieved with excellent rate capabilities. Further, a capacity of 148 mAh g−1 at a current density of 10 A g−1 is obtained with excellent cycling stability, opening a new direction for the fabrication of 3D graphene anodes directly on current collectors for metal ion battery anodes as well as other potential applications.
|
A Hierarchical Three-Dimensional Porous Laser-Scribed Graphene Film for Suppressing Polysulfide Shuttling in Lithium–Sulfur BatteriesThe lithium–sulfur (Li-S) battery is a promising next-generation rechargeable battery with high energy density. Given the outstanding capacities of sulfur (1675 mAh g–1) and lithium metal (3861 mAh g–1), Li-S battery theoretically delivers an ultrahigh energy density of 2567 Wh kg–1. However, this energy density cannot be realized due to several factors, particularly the shuttling of polysulfide intermediates between the cathode and anode, which causes serious degradation of capacity and cycling stability of a Li-S battery. In this work, a simple and scalable route was employed to construct a freestanding laser-scribed graphene (LSG) interlayer that effectively suppresses the polysulfide shuttling in Li-S batteries. Thus, a high specific capacity (1160 mAh g–1) with excellent cycling stability (80.4% capacity retention after 100 cycles) has been achieved due to the unique structure of hierarchical three-dimensional pores in the freestanding LSG.
|
Status and Prospects of Laser-Induced Graphene for Battery ApplicationsSynthesis of 3D graphene by laser irradiation has emerged as a promising approach due to its multifunctionality, cost effectiveness, and simplicity. Herein, the emerging applications of laser-induced graphene (LIG) in batteries are focused on. This type of 3D graphitic carbon offers several advantages, including 1) binder-free self-supported electrode configuration, 2) high electrical and ionic conductivity, 3) hierarchical porosity, and 4) controllable composition upon laser exposure. A comprehensive review of the current status of LIG synthesis and its development for battery applications is discussed. This includes using LIG as an electrode for lithium- and sodium-ion batteries, a current collector for lithium-metal batteries, an electrocatalyst for metal–air batteries, and a host and interlayer for lithium–sulfur batteries. Finally, the article concludes by giving the authors’ perspectives and outlook for developing this class of carbon materials for advanced battery systems.
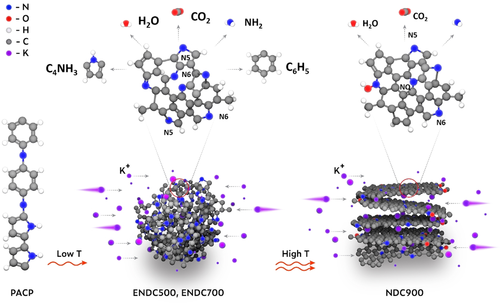
A Site-Selective Doping Strategy of Carbon Anodes with Remarkable K-Ion Storage CapacityThe limited potassium-ion intercalation capacity of graphite hampers development of potassium-ion batteries (PIB). Edge-nitrogen doping is an effective approach to enhance K-ion storage in carbonaceous materials. One shortcoming is the lack of precise control over producing the edge-nitrogen configuration. Here, a molecular-scale copolymer pyrolysis strategy is used to precisely control edge-nitrogen doping in carbonaceous materials. This process results in defect-rich, edge-nitrogen doped carbons (ENDC) with a high nitrogen-doping level (up to 10.5 at %) and a high edge-nitrogen ratio (87.6 %). The optimized ENDC exhibits a high reversible capacity of 423 mAh g−1, a high initial Coulombic efficiency of 65 %, superior rate capability, and long cycle life (93.8 % retention after three months). This strategy can be extended to design other edge-heteroatom-rich carbons through pyrolysis of copolymers for efficient storage of various mobile ions.
|